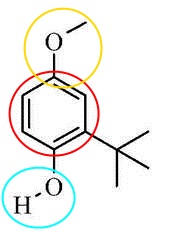
BHA and BHT
BHA and BHT have antioxidant properties meaning that they prevent oxidation reactions.
The hydroxyl group on both chemicals make these molecules more soluble in water as the functional group makes the molecules more polar. However, the ether functional group found on BHA decreases the compounds solubility. Chemicals with benzene rings often give off pleasing smells.
Reactions
These chemicals suppress the oxidation reactions which occur when breaking down fats and oils.
Oxidation reaction for BHA
CH3-O-C6H3(OH)C(CH3)3 + R-O-O → CH3-O-C6H3=(O)C(CH3)3 + R-O-O-H
"R" represents any chain of carbons.
Oxidation reaction for BHT
((CH3)3C)2-CH3-C6H2-OH + R-O-O → ((CH3)3C)2-CH3-C6H2=O + R-O-O-H
"R" represents any chain of carbons.
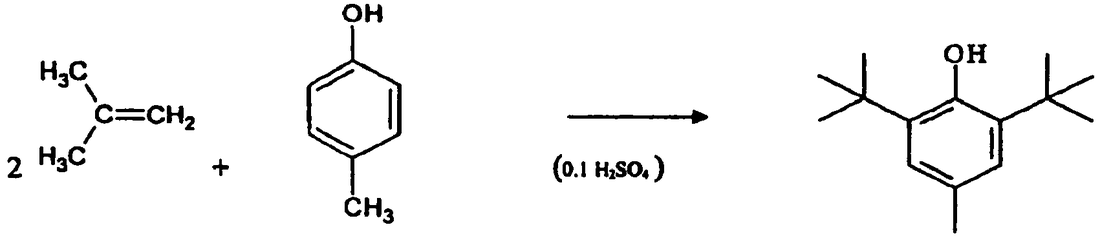
Formation of BTH
2 CH2=C(CH3)2 + CH3(C6H4)OH → ((CH3)3C)2-CH3-C6H2-OH (BHT)
2-methylprop-1-ene + 4-methyl phenol = BHT
Oxidation reaction for BHA
CH3-O-C6H3(OH)C(CH3)3 + R-O-O → CH3-O-C6H3=(O)C(CH3)3 + R-O-O-H
"R" represents any chain of carbons.
Oxidation reaction for BHT
((CH3)3C)2-CH3-C6H2-OH + R-O-O → ((CH3)3C)2-CH3-C6H2=O + R-O-O-H
"R" represents any chain of carbons.
Formation of BTH
2 CH2=C(CH3)2 + CH3(C6H4)OH → ((CH3)3C)2-CH3-C6H2-OH (BHT)
2-methylprop-1-ene + 4-methyl phenol = BHT